-
To prevent/delay progression to T2DM and maintain QOL: Use lifestyle modifications, consider metformin, monitor regularly; discuss DM technology-assisted tools for self-mgmt/support. - Refer adults w/ overweight/obesity at high risk of T2DM (pts w/ IGT, IFG,1 or A1C 5.7%-6.4%2) to intensive lifestyle behavior change program targeting 7% body wt loss and increasing mod-intensity physical activity (e.g., brisk walking) to at least 150min/wk. [A]
- Metformin3,4 to prevent T2DM has the strongest evidence and long-term safety profile. Consider if BMI ≥35 kg/m2, age 25-59 yo, FPG ≥110 mg/dL, A1C ≥6.0%, or hx of gestational DM. [A]
- Variety of eating patterns acceptable for pts w/ prediabetes [B], including Mediterranean and low-carb diets.
- Monitor at least annually [E] for development of T2DM; modify freq based on individual risk/benefit assessment (using informal risk assessment or an assessment tool).
- Screen for and treat modifiable CV risks.5 [B]
- Statin tx may increase risk of T2DM in at-risk pts; monitor glucose status and reinforce DM prevention in such pts. Statin d/c isn’t recommended. [B]
- Stroke/MI prevention: To lower stroke/MI risk, consider pioglitazone in pts w/ stroke hx and evidence of insulin resistance/prediabetes. Benefit needs to be balanced w/ increased risk of wt gain, edema, fx [A]. Lower doses may mitigate ADR risk but may be less effective [C].
- Based on pt preference, consider DM technology-assisted tools to help prevent/delay T2DM onset. [B]
- Consider referral to RDN for individualized medical nutrition tx.
- Obesity mgmt/wt loss: Care goals should include wt loss or prevention of wt gain, minimizing progression of hyperglycemia, and addressing CV risk [B]. Consider pharmacotherapy to support pt-centered goals [B]. Consider more-intensive preventive strategies in pts at very high risk of DM (BMI ≥35 kg/m2, FPG 110-125 mg/dL, 2-hr post-challenge glucose 173-199 mg/dL, A1C ≥6.0%) or hx of gestational DM [A].
- No clear evidence that dietary supplements (e.g., herbs and botanicals, high-dose vitamins and minerals, amino acids, enzymes, antioxidants, etc.) are effective for obesity mgmt/wt loss.6 Supplementation of certain vitamins and minerals (e.g., iron, vit B12, vit D) may be indicated in pts w/ documented def; protein supplements may also be appropriate as adjuncts to medically supervised wt loss regimens.
Footnotes 1 IGT (impaired glucose tolerance) = 2-hr PG 140-199 mg/dL during 75-g OGTT; IFG (impaired fasting glucose) = FPG 100-125 mg/dL.
2 Presence of certain hemoglobinopathies or conditions affecting RBC turnover may affect A1C screen for prediabetes.
3 In addition to metformin, α-glucosidase inhibitors, GLP-1 RAs (liraglutide, semaglutide), TZDs, testosterone, and insulin have been shown to decrease incident T2DM in specific populations.
4 D/t the assoc of long-term metformin use w/ biochemical vit B12 def, consider periodic measurement of vit B12 levels, esp in pts w/ anemia or peripheral neuropathy [B].
5 Prediabetes is assoc w/ heightened CV risk; people w/ prediabetes often have other CV RFs such as HTN and dyslipidemia.
6 Several large systematic reviews show that most trials evaluating dietary supplements for wt loss are of low quality and at high risk for bias. High-quality published studies show little or no wt loss benefits.
-
Awaiting pharmacotherapy approach to glucose lowering General tx considerations - Consider healthy lifestyle behaviors, DM self-mgmt education and support (DSMES), avoidance of therapeutic inertia, and social determinants of health (SDOH) when choosing glucose-lowering tx. [A]
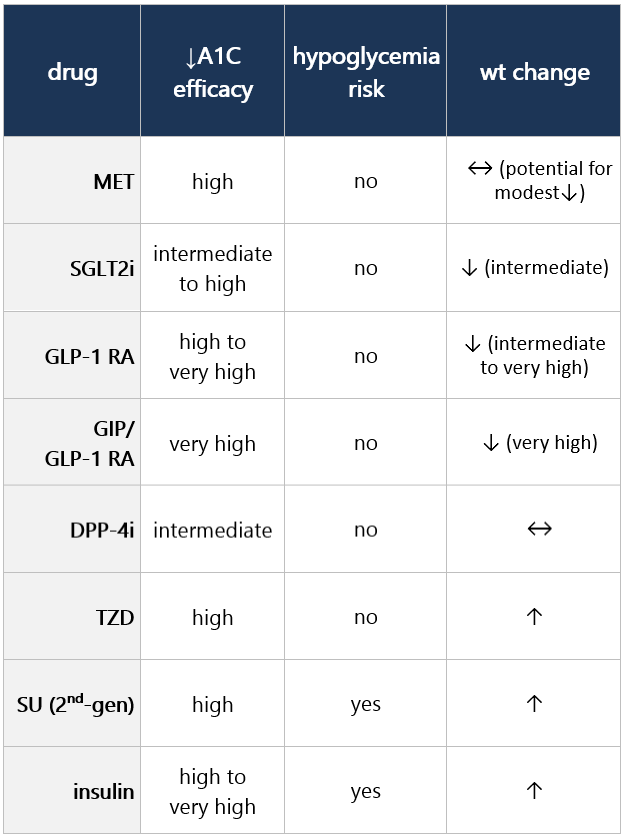
- Use a person-centered shared decision-making approach to guide choice of pharmacologic agents. Consider effects on CV and renal comorbidities; effectiveness; hypoglycemia risk; impact on wt, cost, and access; risk for adverse reactions and tolerability; and individual preferences. [E]
- Glucose-lowering tx plan should consider approaches that support wt mgmt goals. [A]
- Use pharmacological strategies that provide sufficient effectiveness to achieve and maintain intended tx goals. [A]
- Don’t delay tx modification (intensification or deintensification) for adults not meeting individualized tx goals. [A]
- Re-evaluate medication plan and medication-taking behavior at regular intervals (e.g., q3-6mo) and adjust as needed to incorporate specific factors that impact choice of tx. [E]
- Consider early combo tx at tx initiation to shorten time to attainment of individualized tx goals. [A]
- In pts w/o CV and/or kidney dz, pharmacologic agents should address both individualized glycemic and wt goals. [A]
- In pts who haven’t achieved their individualized glycemic goals, selection of subsequent glucose-lowering tx should account for individualized glycemic and wt goals plus presence of other metabolic comorbidities and risk of hypoglycemia. [A]
- In pts who haven’t achieved their individualized weight goals, additional wt mgmt interventions (e.g., intensification of lifestyle modifications, structured wt mgmt programs, pharmacologic agents, or metabolic surgery, as appropriate) are recommended. [A]
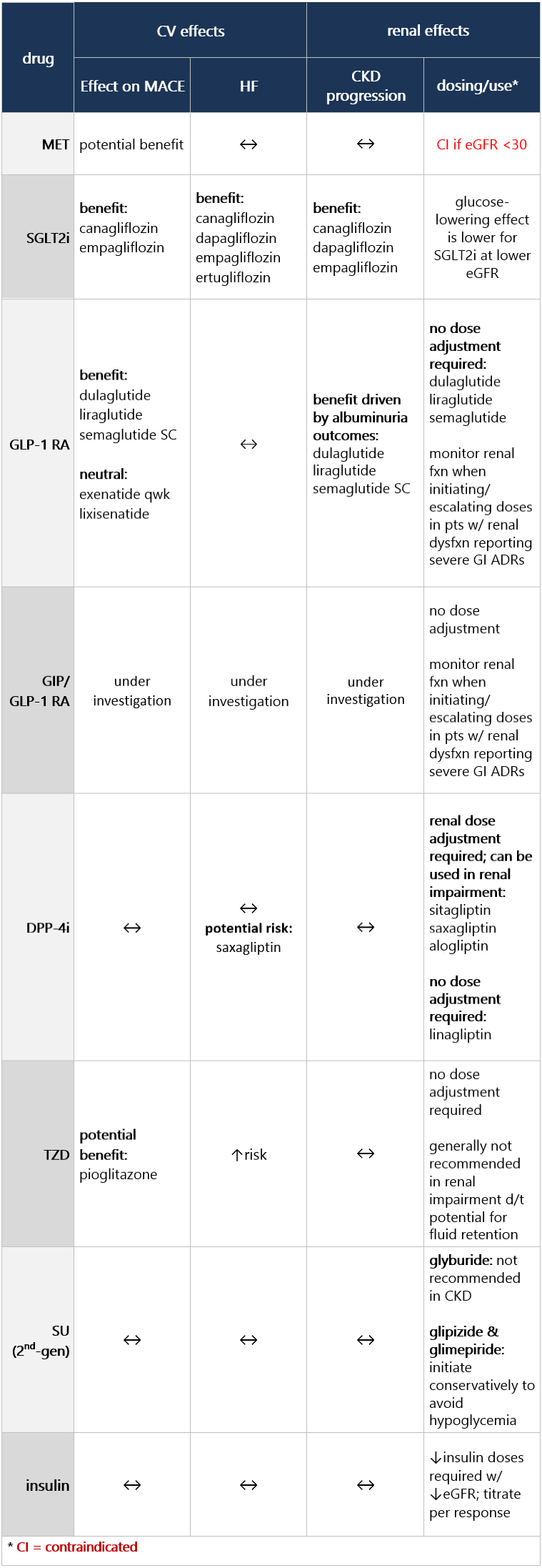
Adults w/ CV or renal dz - In adults w/ T2DM and established or high risk of ASCVD, HF, and/or CKD, tx plan should include agent(s) that reduce(s) CV and kidney dz risk (e.g., SGLT2 and/or GLP-1 RA) for glycemic mgmt and comprehensive CV risk reduction, independent of A1C and in consideration of person-specific factors. [A]
- In pts who have HF (w/ either reduced or preserved EF), an SGLT2 inhibitor is recommended for glycemic mgmt and prevention of HF hospitalizations. [A]
- In pts who have CKD (w/ confirmed eGFR of 20-60 and/or albuminuria), use an SGLT2 inhibitor to minimize progression of CKD and reduce CKD events and HF hospitalizations. Note: Glycemic benefits of SGLT2 inhibitors are reduced at eGFR <45. [A]
- In pts w/ advanced CKD (eGFR <30), a GLP-1 RA is preferred for glycemic mgmt due to lower risk of hypoglycemia and for CV event reduction. [B]
Insulin tx considerations - Consider initiation of insulin regardless of background glucose-lowering tx or dz stage if there’s evidence of ongoing catabolism (e.g., unexpected wt loss), if sx of hyperglycemia are present, or when A1C or blood glucose levels are very high (i.e., A1C >10% or blood glucose ≥300 mg/dL). [E]
- In adults w/ T2DM, a GLP-1 RA, including a dual GIP/GLP-1 RA, is preferred to insulin. [A]
- If insulin is used, combo tx w/ a GLP-1 RA, including a dual GIP/GLP-1 RA, is recommended for greater glycemic effectiveness as well as beneficial effects on wt and hypoglycemia risk. Reassess insulin dosing upon addition or dose escalation of a GLP-1 RA or dual GIP/GLP-1 RA. [A]
- Glucose-lowering agents may be continued upon initiation of insulin tx (unless contraindicated or not tolerated) for ongoing glycemic and metabolic benefits (i.e., wt, cardiometabolic, or kidney benefits). [A]
- To minimize risk of hypoglycemia and tx burden when starting insulin tx, reassess the need for and/or dose of glucose-lowering agents w/ higher hypoglycemia risk (i.e., SUs and meglitinides). [A]
- Monitor for signs of overbasalization during insulin tx, such as basal dose exceeding ∼0.5 units/kg/day, significant bedtime-to-morning or postprandial-to-preprandial glucose differential, occurrences of hypoglycemia (aware or unaware), and high glycemic variability. When overbasalization is suspected, a thorough re-eval should occur promptly to further tailor tx to the individual’s needs. [E]
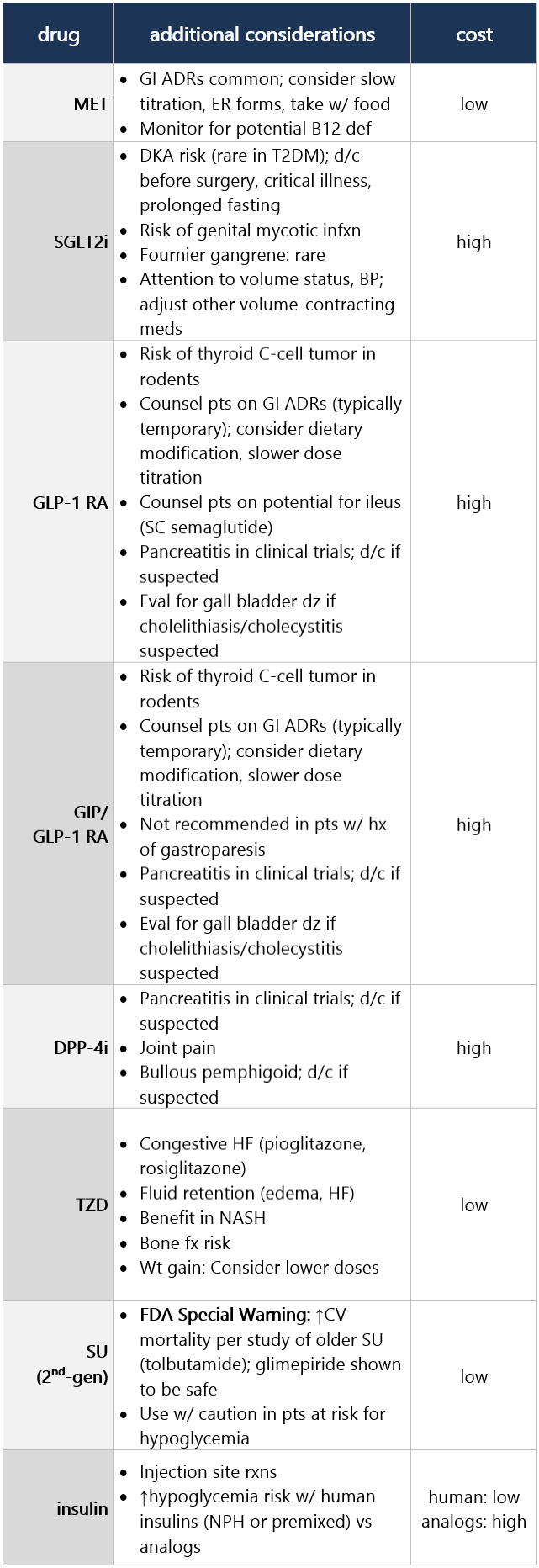
Cost considerations - Routinely assess all pts w/ DM for financial obstacles that could impede their DM mgmt. Clinicians, members of the diabetes care team, and social services professionals should work collaboratively, as appropriate and feasible, to support these individuals by implementing strategies to ↓ costs, thereby improving access to evidence-based care. [E]
- In adults w/ DM and cost-related barriers, consider use of lower-cost medications for glycemic mgmt (i.e., metformin, SUs, TZDs, and human insulin) w/in the context of their risks for hypoglycemia, wt gain, CV and kidney events, and other adverse effects. [E]
Considering trade-off decisions between drug classes (efficacy, wt effects, cost, etc.) Efficacy, hypoglycemia risk, and wt concerns Additional considerations With comorbidity (ASCVD or risk, HF, CKD) Undertake cardiorenal risk redux while considering healthy lifestyle behaviors, DM self-mgmt education and support (DSMES), and social determinants of health (SDOH). - In pts w/ HF, CKD, established CVD, or multiple RFs for CVD, base decision to use a GLP-1 RA or SGLT2i independent of background use of metformin.
- Avoid tx inertia by reassessing/modifying tx regularly (q3-6mo).
Footnotes 1 Established ASCVD warrants strong recommendation, while high-risk indicators warrant a weaker recommendation. Plus, higher absolute risk redux (lower NNT) seen at higher levels of baseline risk and should be factored into decision-making.
2 ASCVD is defined differently across CV outcomes trials, but all included those w/ established CVD (MI, stroke, any revascularization procedure). Variably included conditions like TIA, unstable angina, amputation, symptomatic or asymptomatic CAD.
3 High-risk indicators: Definitions vary, mostly comprise ≥55 yo w/ ≥2 additional RFs (including obesity, smoking, dyslipidemia, albuminuria).
4 For GLP-1 RAs, CV outcomes trials show their efficacy in reducing composite MACE, CV death, all-cause mortality, MI, stroke, and renal endpoints in pts w/ T2DM and established/high risk for CVD.
5 A low-dose TZD may be better tolerated w/ similar efficacy.
6 For SGLT2is, CV/renal trials show their efficacy in reducing risk of composite MACE, CV death, all-cause mortality, MI, hospitalization for HF, and renal outcomes in pts w/ T2DM and established/high risk for CVD.
7 CKD = eGFR <60 mL/min/1.73m2 OR albuminuria (ACR ≥3.0 mg/mmol (30 mg/g)). Since eGFR and albuminuria may vary over time, repeat measurements are needed to document CKD.
8 Use SGLT2i in pts w/ eGFR ≥20 mL/min/1.73m2. Once started, continue until dialysis or transplantation. Without comorbidity: Initial hyperglycemia and wt mgmt Undertake cardiorenal risk redux while considering healthy lifestyle behaviors, DM self-mgmt education and support (DSMES), and social determinants of health (SDOH). - Avoid tx inertia by reassessing/modifying tx q3-6mo.
Intensification to injectable tx needed To meet individual tx goals, use principles from above sections, including reinforcement of behavioral interventions (wt mgmt, physical activity) and provision of DM self-mgmt education and support (DSMES). - Avoid tx inertia by reassessing/modifying tx q3-6mo.
If injectable tx needed to reduce A1C: - Consider GLP-1 RA1 or GIP/GLP-1 RA in most pts prior to insulin initiation. Initiate appropriate start dose (varies by agent w/in class) and gradually titrate to maintenance (varies by agent).
- If already on GLP-1 RA or GIP/GLP-1 RA or if these aren’t appropriate or insulin is preferred, proceed to basal insulin (next step below).
- Consider insulin as the 1st injectable if evidence of ongoing catabolism (wt loss), if hyperglycemia sx are present, if very high A1C levels (>10%) or BG levels (≥300 mg/dL), or if T1DM dx is a possibility.
If A1C still above target, add basal insulin2 (basal analog or bedtime NPH insulin3). - Initiation: Start at 10 units/day OR 0.1-0.2 units/kg/day.
- Titration: Set FPG target; choose evidence-based titration algorithm (e.g., increasing 2 units q3 days to reach FPG target w/o hypoglycemia); for hypoglycemia, determine cause; if no clear reason, lower dose by 10% to 20%.
- Assess adequacy of basal insulin dose: Consider clinical signals to evaluate for overbasalization and need to consider adjunctive tx (e.g., basal dose >0.5 units/kg/day, elevated bedtime-morning and/or postprandial differential, hypoglycemia (aware or unaware), high variability).
If above A1C target and not already on GLP-1 RA or GIP/GLP-1 RA, consider these classes, either in free combo or fixed-ratio combo, w/ insulin. If A1C still above target, add prandial insulin (or if on bedtime NPH, consider switch to bid NPH regimen4). - Usually 1 dose w/ largest meal or meal w/ greatest PPG excursion; prandial insulin can be dosed individually or mixed w/ NPH.
- Initiation: 4 units/day or 10% of basal dose. If A1C <8%, consider lowering basal dose by 4 units or 10% of basal dose.
- Titration: Increase dose by 1-2 units or 10% to 15% twice weekly; for hypoglycemia, determine cause; if no reason, lower corresponding dose by 10% to 20%.
If A1C still above target, consider 1 of 3 options: - Stepwise additional injections of prandial insulin (i.e., 2, then 3, injections); then proceed to full basal-bolus regimen (i.e., basal insulin and prandial insulin w/ each meal).
- Self-mixed/split insulin regimen (can adjust NPH and short/rapid-acting insulins separately)5
- Twice-daily premix insulin regimen6
Footnotes 1 When selecting GLP-1 RA, consider pt preference, A1C lowering, wt-lowering effect, or freq of injection. If CVD: Consider GLP-1 RA w/ proven CVD benefit. PO or injectable GLP-1 RA is appropriate.
2 For pts on GLP-1 RA and basal insulin combo, consider fixed-ratio combo product (IDegLira or iGlarLixi).
3 Consider switch from evening NPH to basal analog if pt develops hypoglycemia and/or frequently forgets to administer NPH in the evening and would be better managed w/ morning dose of long-acting basal insulin.
4 Conversion of bedtime NPH to bid NPH based on individual needs and current glycemic control. One possible approach:
• Initiation: Total dose = 80% of current NPH dose; 2/3 given in AM, 1/3 in PM.
• Titration: based on individual needs.
• If still above target A1C, proceed to prandial insulin.
5 Self-mixed/split insulin regimen:
• Initiation: Total NPH dose = 80% of current NPH dose; 2/3 given before breakfast, 1/3 given before dinner; add 4 units of short/rapid-acting insulin to each injection or 10% of reduced NPH dose.
• Titration: Titrate each component of regimen based on individual needs.
6 Twice-daily premix insulin regimen:
• Initiation: usually unit per unit at same total insulin dose but may require adjustment to individual needs.
• Titration: based on individual needs.
|