| Guideline | Resources |
| Periprocedural Management of Oral Anticoagulants | 2023 ACC/AHA/ACCP/HRS, 2017 ACC Guideline Synthesis |
Key Points
Aim is to hold OAC for shortest time to minimize risk of thromboembolism and to facilitate hemostasis and avoid intraoperative and postoperative bleeding. In all pts, evaluate the bleeding risk of the procedure, pt-specific risk factors, and thrombotic risks while off OACs. Risk of thromboembolism during temporary stop of OAC is generally 1%. These recommendations apply to elective, not urgent/emergent, procedures.
Choose Patient Type
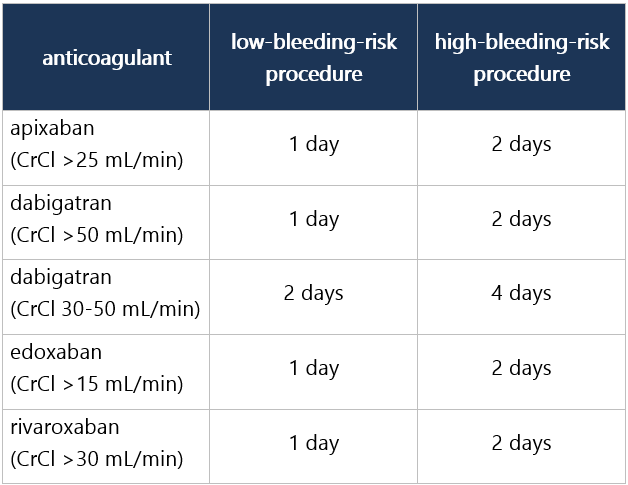
|
Peer reviewed & based on multiple guidelines/recommendations from specialty societies/government agencies.
Epocrates Guideline Synopsis Last Update:
Mar 26, 2024
Publication Year:
2023
Source:
epocrates
Levels of Evidence
| [A] | High-quality evidence from ≥1 RCT, or ≥1 RCT corroborated by high-quality registries |
| [B-R] | Mod-quality evidence from ≥1 RCT |
| [B-NR] | Mod-quality evidence from ≥1 well-designed, well-executed nonrandomized, observational, or registry study |
| [C-LD] | Observational/registry studies w/ limitations of design/execution; physiological/mechanistic studies |
| [C-EO] | Consensus of expert opinion based on clinical experience |
Abbreviations
| AF | atrial fibrillation |
| CrCl | creatinine clearance |
| DOAC | direct oral anticoagulant |
| EtOH | alcohol |
| INR | international normalized ratio |
| LMWH | low-molecular-weight heparin |
| NSAID | nonsteroidal anti-inflammatory drug |
| OAC | oral anticoagulant |
| TIA | transient ischemic attack |
Levels of Evidence
| A | = | High-quality evidence from ≥1 RCT, or ≥1 RCT corroborated by high-quality registries |
| B-R | = | Mod-quality evidence from ≥1 RCT |
| B-NR | = | Mod-quality evidence from ≥1 well-designed, well-executed nonrandomized, observational, or registry study |
| C-LD | = | Observational/registry studies w/ limitations of design/execution; physiological/mechanistic studies |
| C-EO | = | Consensus of expert opinion based on clinical experience |