-
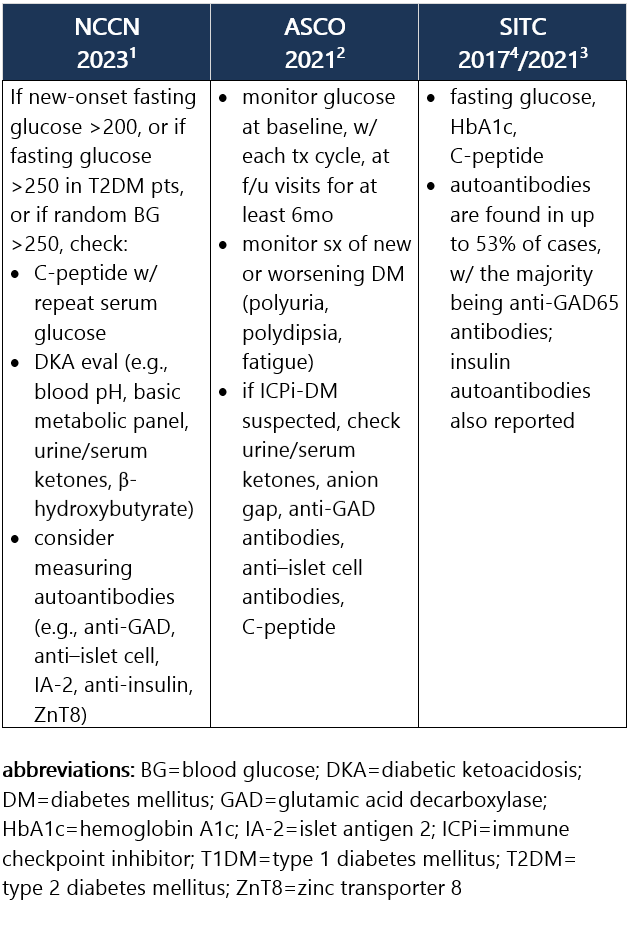
ICPi-associated type 1 diabetes mellitus (ICPi-T1DM) is rare and often presents as life-threatening fulminant T1DM and DKA initially. However, pts may also be asymptomatic or present w/ acute onset of polyuria, polydipsia, wt loss, and lethargy. Diabetes may occur shortly after the start of ICPi tx or as much as 1y following the start of tx, at an incidence of ~1%.1-3 Recommendations for pt eval: View checkpoint inhibitors drug info: Footnotes 1 NCCN 2023. Thompson JA, et al. Management of Immunotherapy-Related Toxicities, Version 1.2023, NCCN Clinical Practice Guidelines in Oncology. Epub 2023 Mar 10. Accessed 04/24/2023.
2 ASCO 2021. Schneider BJ, et al. Management of Immune-Related Adverse Events in Patients Treated With Immune Checkpoint Inhibitor Therapy: ASCO Guideline Update. J Clin Oncol. 2021 Dec 20;39(36):4073-4126. Accessed 04/20/2023.
3 SITC 2021. Brahmer JR, et al. Society for Immunotherapy of Cancer (SITC) Clinical Practice Guideline on Immune Checkpoint Inhibitor-Related Adverse Events. J Immunother Cancer. 2021 Jun;9(6):e002435. Free full-text PDF at PubMed® Central. Accessed 04/20/2023.
4 SITC 2017. Puzanov I, et al. Managing Toxicities Associated with Immune Checkpoint Inhibitors: Consensus Recommendations from the Society for Immunotherapy of Cancer (SITC) Toxicity Management Working Group. J Immunother Cancer. 2017 Nov 21;5(1):95. Free full-text PDF at PubMed® Central. Accessed 04/20/2023.
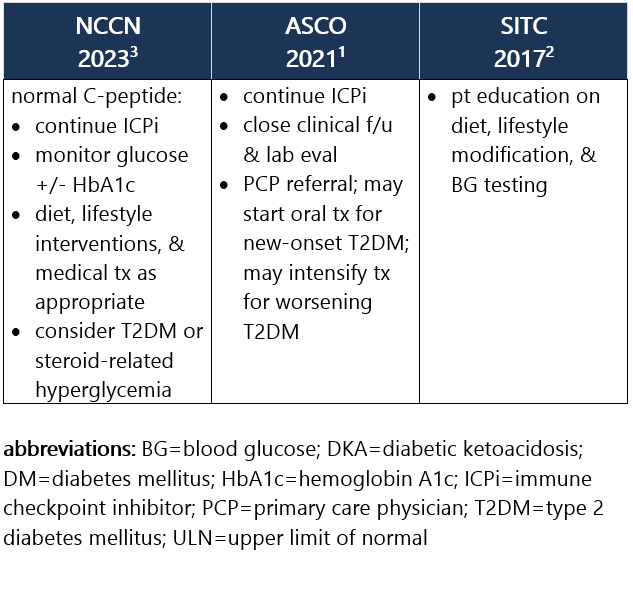
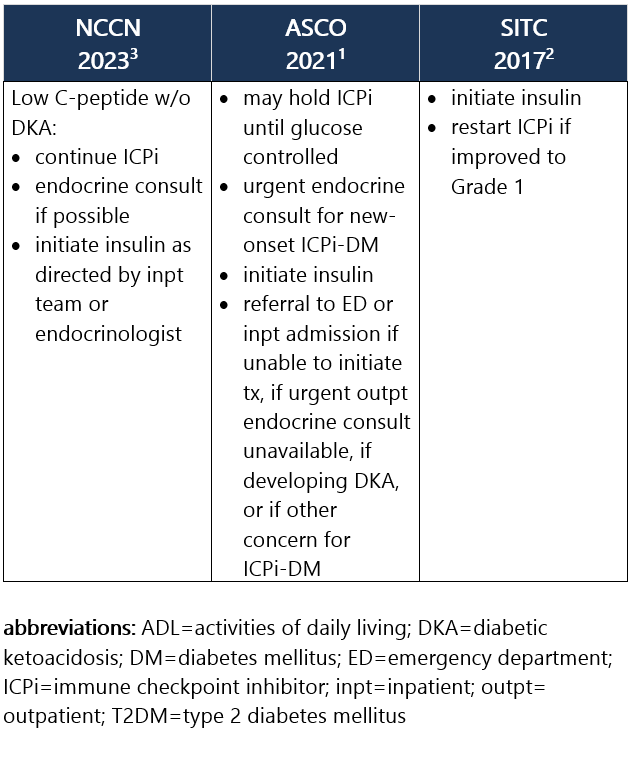
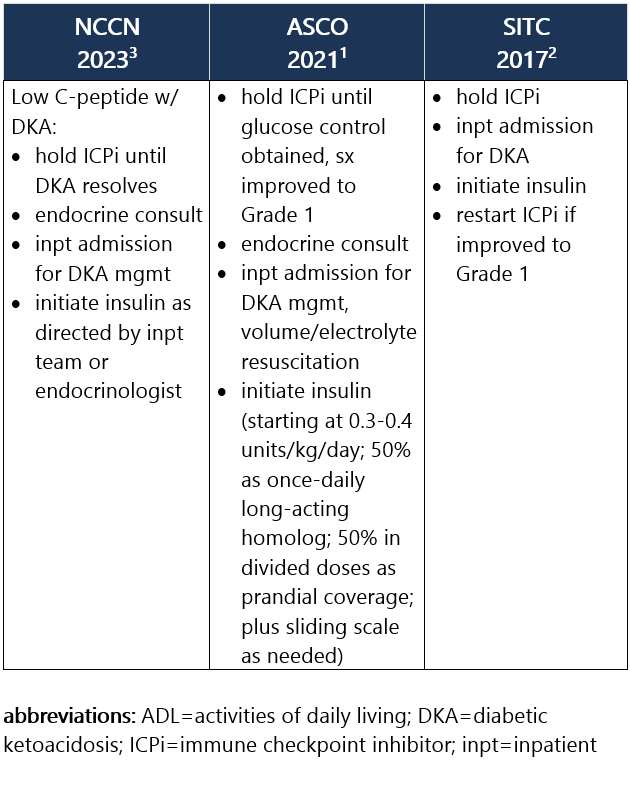
Grade 1 – Asymptomatic or mild sx; T2DM w/ fasting glucose >ULN to 160 mg/dL (>ULN to 8.9 mmol/L); no evidence of ICPi-DM, DKA, or pancreatic autoimmunity1,2 Management recommendations: Grade 2 – Moderate sx, able to perform ADL; T2DM w/ fasting glucose >160 to 250 mg/dL (>8.9 to 13.9 mmol/L); no DKA or metabolic derangement but other evidence of ICPi-DM at any glucose level1,2 Management recommendations: Grade 3 – Severe sx, medically significant or life-threatening consequences, unable to perform ADL; fasting glucose >250 to 500 mg/dL (>13.9 to 27.8 mmol/L); DKA or other metabolic abnormality1,2 Management recommendations: Grade 4 – Severe sx, medically significant or life-threatening consequences, unable to perform ADL; fasting glucose >500 mg/dL (>27.8 mmol/L); DKA or other metabolic abnormality1,2 Management recommendations: View checkpoint inhibitors drug info: Footnotes 1 ASCO 2021. Schneider BJ, et al. Management of Immune-Related Adverse Events in Patients Treated With Immune Checkpoint Inhibitor Therapy: ASCO Guideline Update. J Clin Oncol. 2021 Dec 20;39(36):4073-4126. Accessed 04/20/2023.
2 SITC 2017. Puzanov I, et al. Managing Toxicities Associated with Immune Checkpoint Inhibitors: Consensus Recommendations from the Society for Immunotherapy of Cancer (SITC) Toxicity Management Working Group. J Immunother Cancer. 2017 Nov 21;5(1):95. Free full-text PDF at PubMed® Central. Accessed 04/20/2023.
3 NCCN 2023. Thompson JA, et al. Management of Immunotherapy-Related Toxicities, Version 1.2023, NCCN Clinical Practice Guidelines in Oncology. Epub 2023 Mar 10. Accessed 04/24/2023. -
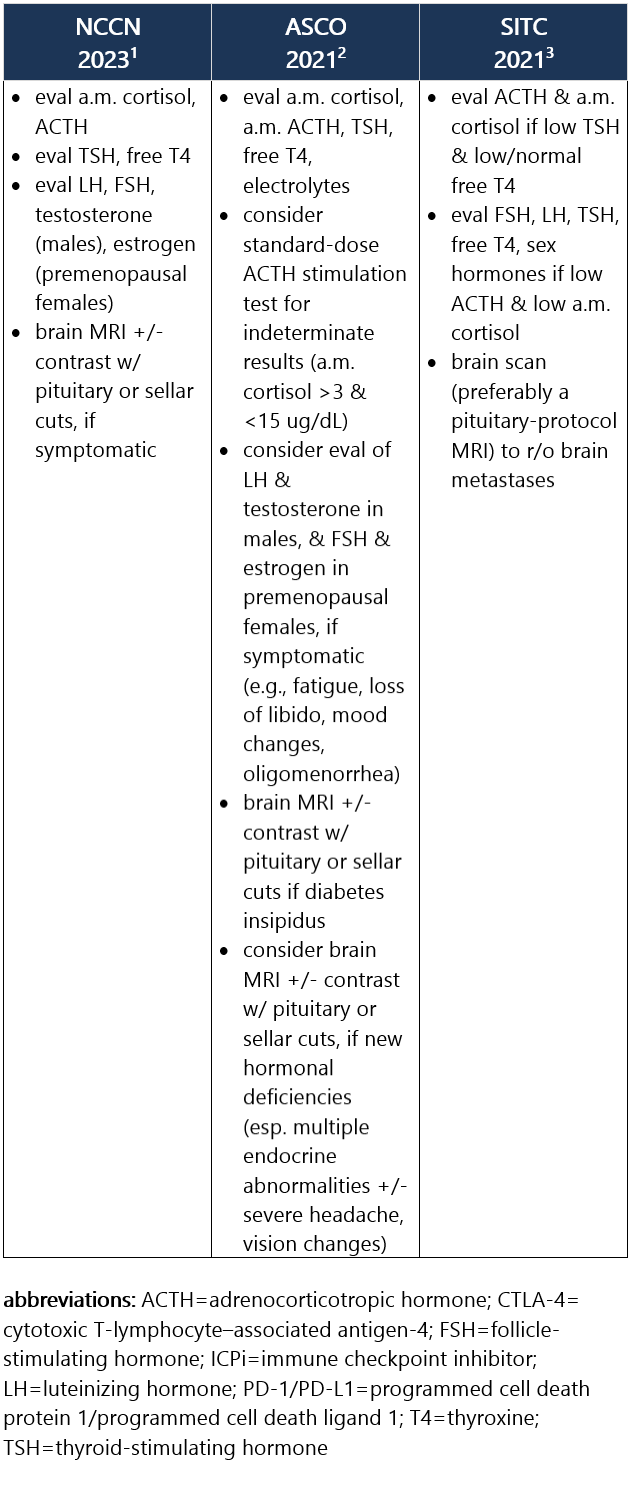
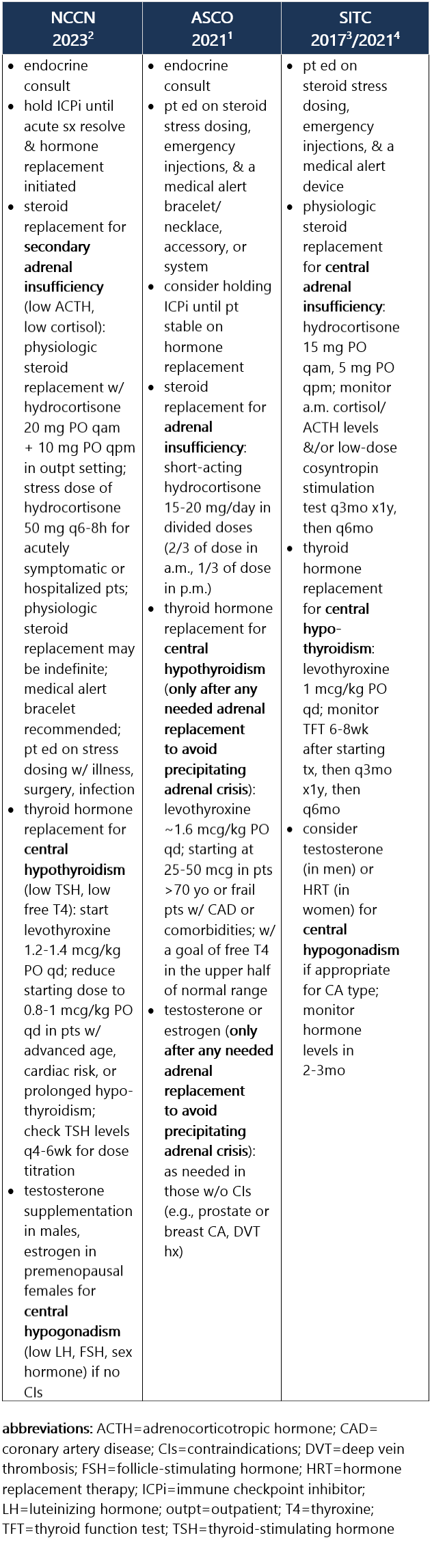
ICPi-related hypophysitis most frequently manifests as secondary (central) adrenal insufficiency d/t ACTH deficiency (less commonly d/t TSH, FSH, LH deficiency). Test results indicative of hypophysitis may show low levels of ACTH, morning cortisol, TSH, free T4, sodium, potassium, testosterone, etc. Median time between ICPi initiation to hypophysitis dx is 76 days. Hypophysitis occurs in 1% of pts treated w/ anti–PD-1/PD-L1 ICPi, in 4% of pts treated w/ anti–CTLA-4 ICPi, and in 9% to 11% of pts treated w/ combo ICPi.1-3 Recommendations for pt eval: View checkpoint inhibitors drug info: Footnotes 1 NCCN 2023. Thompson JA, et al. Management of Immunotherapy-Related Toxicities, Version 1.2023, NCCN Clinical Practice Guidelines in Oncology. Epub 2023 Mar 10. Accessed 04/24/2023.
2 ASCO 2021. Schneider BJ, et al. Management of Immune-Related Adverse Events in Patients Treated With Immune Checkpoint Inhibitor Therapy: ASCO Guideline Update. J Clin Oncol. 2021 Dec 20;39(36):4073-4126. Accessed 04/20/2023.
3 SITC 2021. Brahmer JR, et al. Society for Immunotherapy of Cancer (SITC) Clinical Practice Guideline on Immune Checkpoint Inhibitor-Related Adverse Events. J Immunother Cancer. 2021 Jun;9(6):e002435. Free full-text PDF at PubMed® Central. Accessed 04/20/2023. Grade 1 – Asymptomatic or mild sx1 Management recommendations: Grade 2 – Moderate sx, able to perform ADL1 Management recommendations: Grade 3/4 – Severe sx, medically significant or life-threatening consequences, unable to perform ADL1 Management recommendations: View checkpoint inhibitors drug info: Footnotes 1 ASCO 2021. Schneider BJ, et al. Management of Immune-Related Adverse Events in Patients Treated With Immune Checkpoint Inhibitor Therapy: ASCO Guideline Update. J Clin Oncol. 2021 Dec 20;39(36):4073-4126. Accessed 04/20/2023.
2 NCCN 2023. Thompson JA, et al. Management of Immunotherapy-Related Toxicities, Version 1.2023, NCCN Clinical Practice Guidelines in Oncology. Epub 2023 Mar 10. Accessed 04/24/2023.
3 SITC 2017. Puzanov I, et al. Managing Toxicities Associated with Immune Checkpoint Inhibitors: Consensus Recommendations from the Society for Immunotherapy of Cancer (SITC) Toxicity Management Working Group. J Immunother Cancer. 2017 Nov 21;5(1):95. Free full-text PDF at PubMed® Central. Accessed 04/20/2023.
4 SITC 2021. Brahmer JR, et al. Society for Immunotherapy of Cancer (SITC) Clinical Practice Guideline on Immune Checkpoint Inhibitor-Related Adverse Events. J Immunother Cancer. 2021 Jun;9(6):e002435. Free full-text PDF at PubMed® Central. Accessed 04/20/2023. -
Primary Adrenal Insufficiency
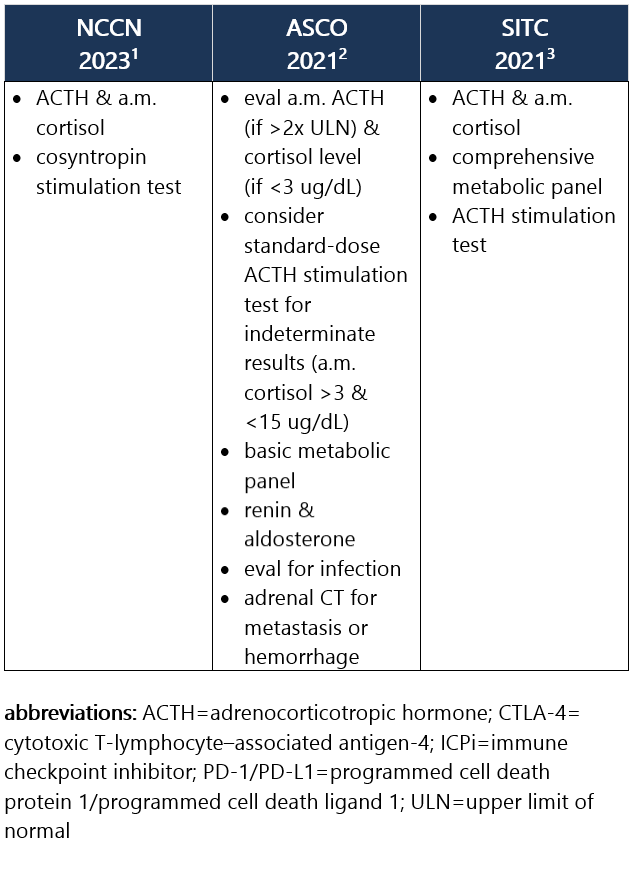
ICPi-related primary adrenal insufficiency is caused by direct damage to the adrenal glands and is associated w/ high ACTH levels along w/ low morning cortisol. As w/ secondary adrenal insufficiency, it can lead to life-threatening adrenal crisis d/t vasodilatory shock. Sx can be nonspecific, including nausea, loss of appetite, wt loss, fatigue, light-headedness, hypoglycemia, and hypotension. It occurs in 1% of pts treated w/ anti–PD-1/PD-L1 ICPi or anti–CTLA-4 ICPi, and in 5% of pts treated w/ combo ICPi, w/ a median onset of 10wk from ICPi initiation.1-3 Recommendations for pt eval: View checkpoint inhibitors drug info: Footnotes 1 NCCN 2023. Thompson JA, et al. Management of Immunotherapy-Related Toxicities, Version 1.2023, NCCN Clinical Practice Guidelines in Oncology. Epub 2023 Mar 10. Accessed 04/24/2023.
2 ASCO 2021. Schneider BJ, et al. Management of Immune-Related Adverse Events in Patients Treated With Immune Checkpoint Inhibitor Therapy: ASCO Guideline Update. J Clin Oncol. 2021 Dec 20;39(36):4073-4126. Accessed 04/20/2023.
3 SITC 2021. Brahmer JR, et al. Society for Immunotherapy of Cancer (SITC) Clinical Practice Guideline on Immune Checkpoint Inhibitor-Related Adverse Events. J Immunother Cancer. 2021 Jun;9(6):e002435. Free full-text PDF at PubMed® Central. Accessed 04/20/2023. Grade 1 – Asymptomatic or mild sx1 Management recommendations: Grade 2 – Moderate sx, able to perform ADL1 Management recommendations: Grade 3/4 – Severe sx, medically significant or life-threatening consequences, unable to perform ADL1 Management recommendations: View checkpoint inhibitors drug info: Footnotes 1 ASCO 2021. Schneider BJ, et al. Management of Immune-Related Adverse Events in Patients Treated With Immune Checkpoint Inhibitor Therapy: ASCO Guideline Update. J Clin Oncol. 2021 Dec 20;39(36):4073-4126. Accessed 04/20/2023.
2 NCCN 2023. Thompson JA, et al. Management of Immunotherapy-Related Toxicities, Version 1.2023, NCCN Clinical Practice Guidelines in Oncology. Epub 2023 Mar 10. Accessed 04/24/2023.
3 SITC 2021. Brahmer JR, et al. Society for Immunotherapy of Cancer (SITC) Clinical Practice Guideline on Immune Checkpoint Inhibitor-Related Adverse Events. J Immunother Cancer. 2021 Jun;9(6):e002435. Free full-text PDF at PubMed® Central. Accessed 04/20/2023. -
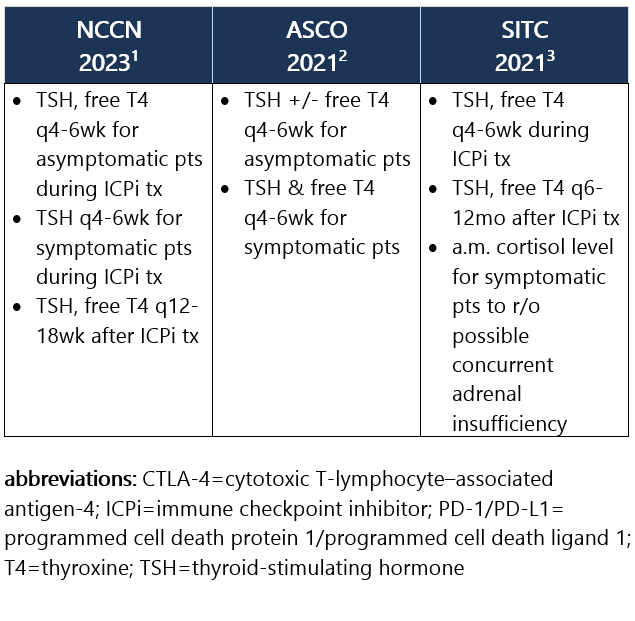
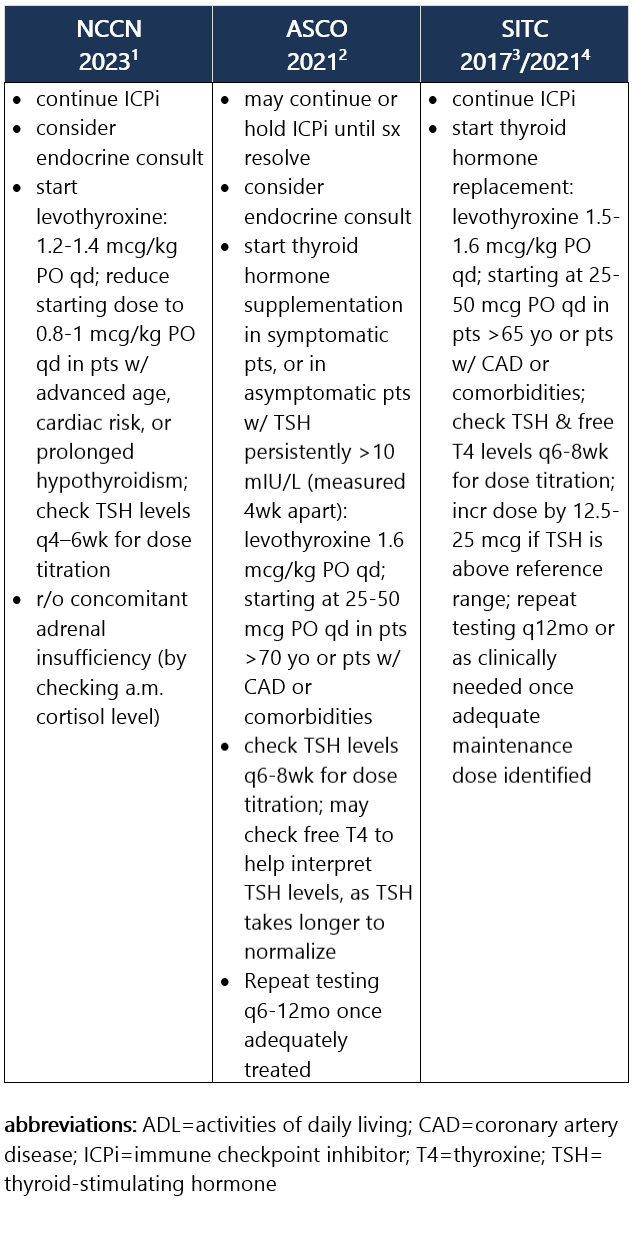
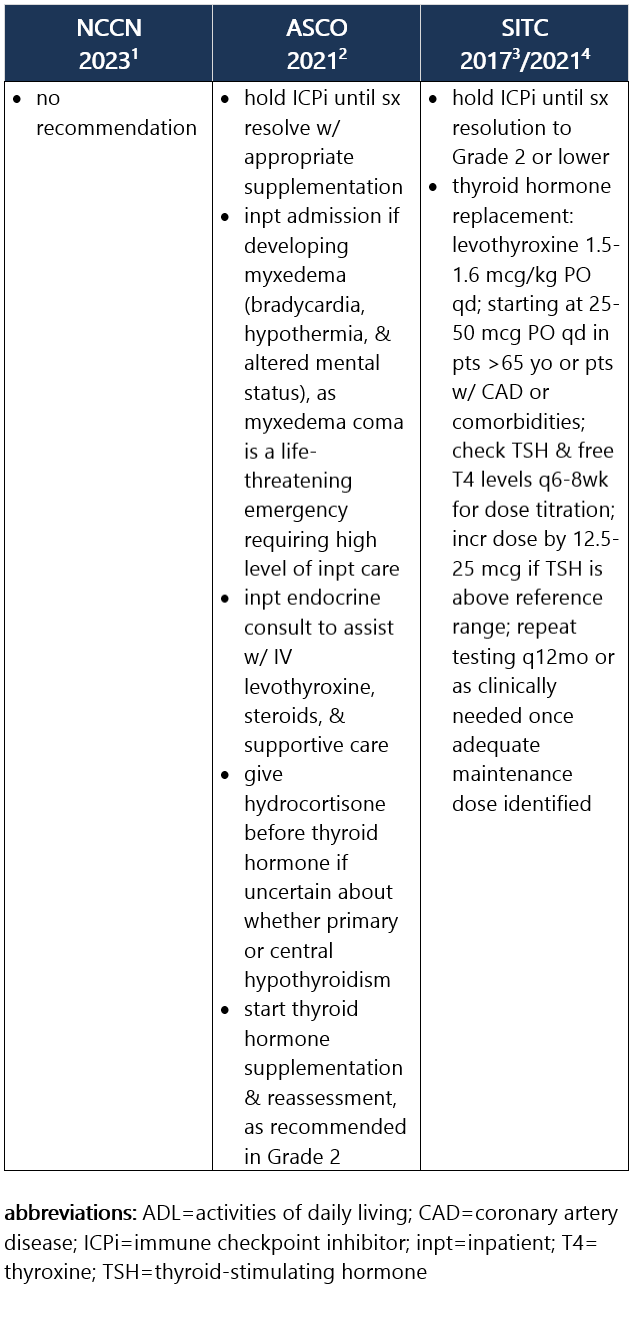
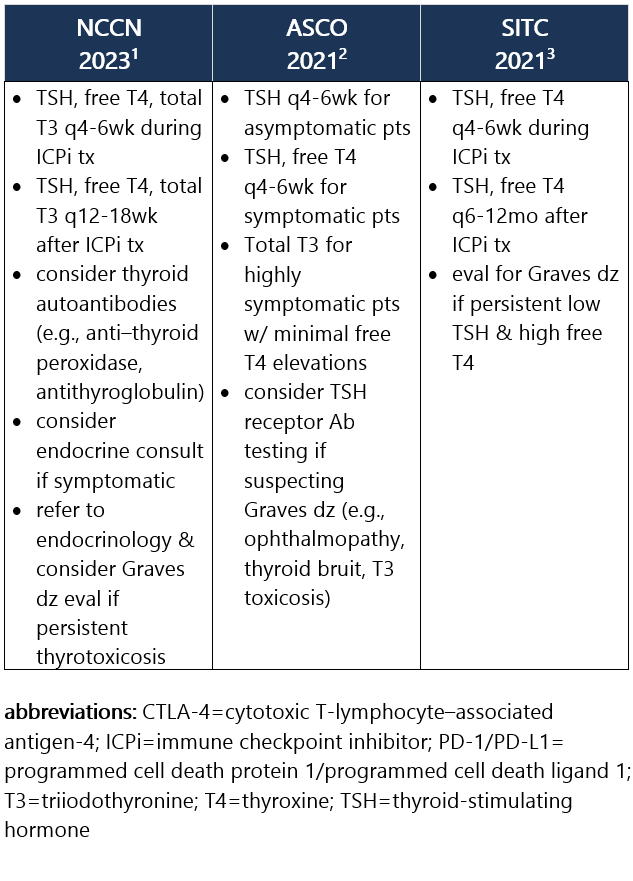
Elevated TSH w/ normal or low free T4. Hypothyroidism is frequently asymptomatic, therefore requiring routine monitoring of TSH and free T4 levels during tx. It commonly develops after the thyrotoxicosis phase of thyroiditis, d/t the destruction of thyroid follicles and necrosis. Median time to hypothyroidism is 10wk after ICPi initiation. Hypothyroidism occurs in ~8% of pts receiving anti–PD-1/PD-L1 ICPi, in 3% of pts receiving anti–CTLA-4 ICPi, and in 15% of pts treated w/ combo ICPi.1-3 Recommendations for pt eval: View checkpoint inhibitors drug info: Footnotes 1 NCCN 2023. Thompson JA, et al. Management of Immunotherapy-Related Toxicities, Version 1.2023, NCCN Clinical Practice Guidelines in Oncology. Epub 2023 Mar 10. Accessed 04/24/2023.
2 ASCO 2021. Schneider BJ, et al. Management of Immune-Related Adverse Events in Patients Treated With Immune Checkpoint Inhibitor Therapy: ASCO Guideline Update. J Clin Oncol. 2021 Dec 20;39(36):4073-4126. Accessed 04/20/2023.
3 SITC 2021. Brahmer JR, et al. Society for Immunotherapy of Cancer (SITC) Clinical Practice Guideline on Immune Checkpoint Inhibitor-Related Adverse Events. J Immunother Cancer. 2021 Jun;9(6):e002435. Free full-text PDF at PubMed® Central. Accessed 04/20/2023. Grade 1 – TSH >4 and <10 mIU/L, normal free T4, and asymptomatic1,2 Management recommendations: Grade 2 – Moderate sx, able to perform ADL. TSH persistently >10 mIU/L1,2 Management recommendations: Grade 3/4 – Severe sx, medically significant or life-threatening consequences, unable to perform ADL2 Management recommendations: View checkpoint inhibitors drug info: Footnotes 1 NCCN 2023. Thompson JA, et al. Management of Immunotherapy-Related Toxicities, Version 1.2023, NCCN Clinical Practice Guidelines in Oncology. Epub 2023 Mar 10. Accessed 04/24/2023.
2 ASCO 2021. Schneider BJ, et al. Management of Immune-Related Adverse Events in Patients Treated With Immune Checkpoint Inhibitor Therapy: ASCO Guideline Update. J Clin Oncol. 2021 Dec 20;39(36):4073-4126. Accessed 04/20/2023.
3 SITC 2017. Puzanov I, et al. Managing Toxicities Associated with Immune Checkpoint Inhibitors: Consensus Recommendations from the Society for Immunotherapy of Cancer (SITC) Toxicity Management Working Group. J Immunother Cancer. 2017 Nov 21;5(1):95. Free full-text PDF at PubMed® Central. Accessed 04/20/2023.
4 SITC 2021. Brahmer JR, et al. Society for Immunotherapy of Cancer (SITC) Clinical Practice Guideline on Immune Checkpoint Inhibitor-Related Adverse Events. J Immunother Cancer. 2021 Jun;9(6):e002435. Free full-text PDF at PubMed® Central. Accessed 04/20/2023. -
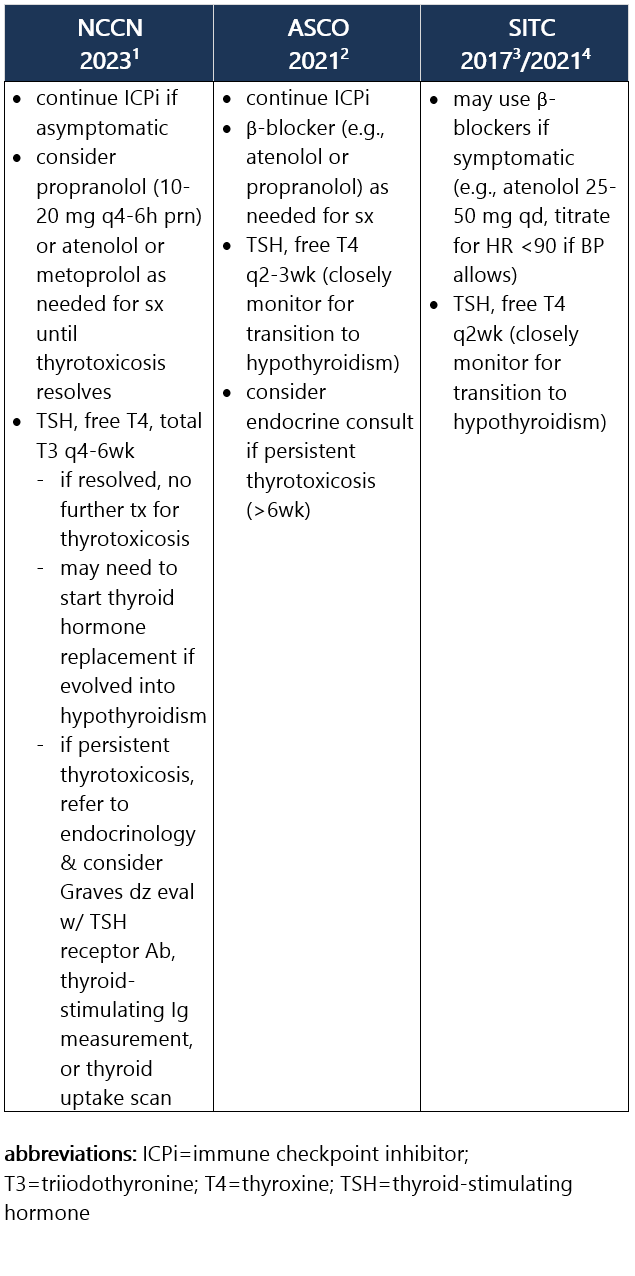
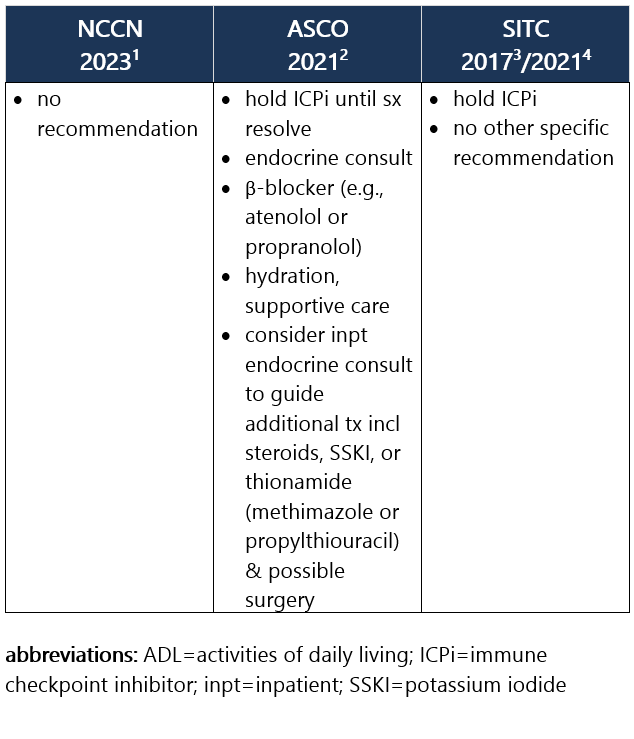
Defined as low or suppressed TSH (<0.01 mIU/L) due to thyroiditis, w/ high free T4 (and/or high total T3). Most pts w/ thyrotoxicosis are asymptomatic. If present, sx may include palpitations, heat intolerance, restlessness or anxiety, fine tremor, and/or wt loss. Thyrotoxicosis occurs in ~5% of pts receiving anti–PD-1/PD-L1 ICPi and in 4% of pts receiving anti–CTLA-4 ICPi, and rarely may lead to Graves dz. Approximately 50% to 90% of thyrotoxicosis cases evolve into hypothyroidism, requiring long-term levothyroxine replacement. Median time to thyrotoxicosis is 5wk after ICPi initiation; usual duration is 4-6wk.1-3 Recommendations for pt eval: View checkpoint inhibitors drug info: Footnotes 1 NCCN 2023. Thompson JA, et al. Management of Immunotherapy-Related Toxicities, Version 1.2023, NCCN Clinical Practice Guidelines in Oncology. Epub 2023 Mar 10. Accessed 04/24/2023.
2 ASCO 2021. Schneider BJ, et al. Management of Immune-Related Adverse Events in Patients Treated With Immune Checkpoint Inhibitor Therapy: ASCO Guideline Update. J Clin Oncol. 2021 Dec 20;39(36):4073-4126. Accessed 04/20/2023.
3 SITC 2021. Brahmer JR, et al. Society for Immunotherapy of Cancer (SITC) Clinical Practice Guideline on Immune Checkpoint Inhibitor-Related Adverse Events. J Immunother Cancer. 2021 Jun;9(6):e002435. Free full-text PDF at PubMed® Central. Accessed 04/20/2023. Grade 1 – Asymptomatic or mild sx1,2 Management recommendations: Grade 2 – Moderate sx, able to perform ADL1,2 Management recommendations: Grade 3/4 – Severe sx, medically significant or life-threatening consequences, unable to perform ADL2 Management recommendations: View checkpoint inhibitors drug info: Footnotes 1 NCCN 2023. Thompson JA, et al. Management of Immunotherapy-Related Toxicities, Version 1.2023, NCCN Clinical Practice Guidelines in Oncology. Epub 2023 Mar 10. Accessed 04/24/2023.
2 ASCO 2021. Schneider BJ, et al. Management of Immune-Related Adverse Events in Patients Treated With Immune Checkpoint Inhibitor Therapy: ASCO Guideline Update. J Clin Oncol. 2021 Dec 20;39(36):4073-4126. Accessed 04/20/2023.
3 SITC 2017. Puzanov I, et al. Managing Toxicities Associated with Immune Checkpoint Inhibitors: Consensus Recommendations from the Society for Immunotherapy of Cancer (SITC) Toxicity Management Working Group. J Immunother Cancer. 2017 Nov 21;5(1):95. Free full-text PDF at PubMed® Central. Accessed 04/20/2023.
4 SITC 2021. Brahmer JR, et al. Society for Immunotherapy of Cancer (SITC) Clinical Practice Guideline on Immune Checkpoint Inhibitor-Related Adverse Events. J Immunother Cancer. 2021 Jun;9(6):e002435. Free full-text PDF at PubMed® Central. Accessed 04/20/2023.
|