-
Patient undergoing dental/oral surgery procedure involving gingival tissue/periapical region manipulation or oral mucosal perforation
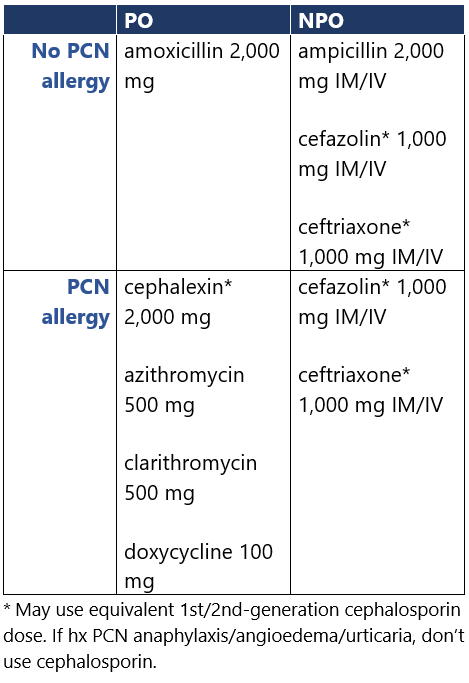
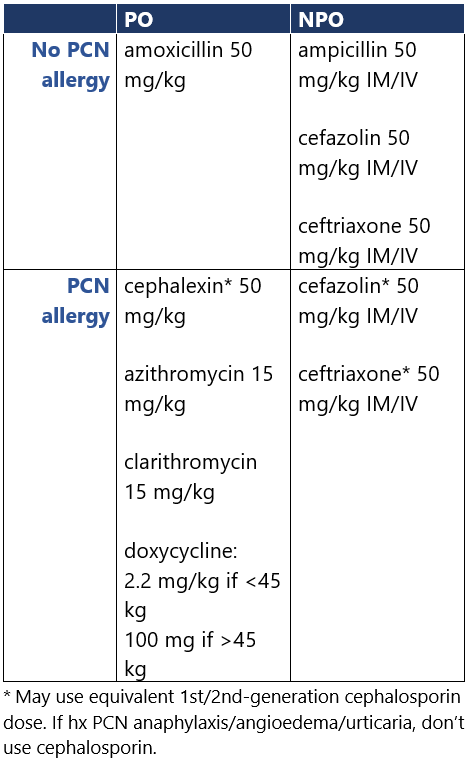
Relevant cardiac condition: 1) prosthetic valve/material, 2) previous IE, 3) cardiac txp w/ valvulopathy, 4) CHD (unrepaired cyanotic dz, repaired defect w/in 6mo, w/ residual defects at site of/adjacent to prosthetic patch/device, or w/ surgical/transcatheter PA valve/conduit) Use antibiotic prophylaxis for pts w/ relevant cardiac conditions.1-3 Give single dose 30-60min before procedure.1,2 If inadvertently not given pre-procedure, give up to 2h after procedure. For repeated dental/oral surgery procedures, vary regimen each time or wait ≥4wk between procedures.1 Guidance from groups differs: - ESC2 suggests considering (rather than recommending) prophylaxis for pts w/ transcatheter mitral/tricuspid valve repair or cardiac txp.
- JCS3 includes clindamycin as an option for pts who are allergic to β-lactams or unable to take oral meds, while AHA, ESC don’t (C. diff risk). JCS doesn’t address cardiac txp; they don’t recommend prophylaxis for dental pulpectomy.
- NICE.4 Antibiotic prophylaxis not routinely recommended for dental procedures; don’t offer chlorhexidine mouthwash as prophylaxis.
Footnotes 1 AHA 2021. Wilson WR, et al. Prevention of Viridans Group Streptococcal Infective Endocarditis: A Scientific Statement From the American Heart Association. Circulation. 2021 May 18;143(20):e963-e978. Full-text article
Four conditions warrant prophylaxis for specific dental/oral surgery procedures:
• Prosthetic cardiac valve/material
◦ Prosthetic valve (including transcatheter-implanted)
◦ Valve repair w/ device (e.g., annuloplasty ring/clip)
◦ LVAD or implantable heart
• Previous/relapsed/recurrent IE
• CHD
◦ Unrepaired cyanotic congenital CHD (including palliative shunt, conduit)
◦ Congenital defect repaired w/ prosthetic material/device (via surgery/transcatheter) w/in 6mo after repair
◦ Repaired w/ residual defects @ site of/adjacent to prosthetic patch/device
◦ Surgical/transcatheter PA valve/conduit
• Cardiac txp w/ valvulopathy
Prophylaxis not recommended for pacemaker/similar implanted electronics, completed closed septal defect devices, peripheral vascular graft/patch (including for HD), coronary/vascular stents, CNS ventriculoatrial shunt, vena cava filter, pledget.
Dental procedures not warranting prophylaxis include x-rays; anesthetic injection in noninfected tissue; placing removable prosthodontic/orthodontic appliances or orthodontic brackets; orthodontic appliance adjustment; primary teeth shedding or lip/oral mucosa traumatic bleeding.
2 ESC 2023. Delgado V, et al. 2023 ESC Guidelines for the management of endocarditis: Developed by the task force on the management of endocarditis of the European Society of Cardiology (ESC). Eur Heart J. Preprint. Posted online August 25, 2023.
Full-text article
3 JCS 2017. Nakatani S, et al. JCS 2017 Guideline on Prevention and Treatment of Infective Endocarditis. Circ J. 2019 Jul 25;83(8):1767-1809.
Full-text article
4 NICE 2016. Prophylaxis against infective endocarditis: antimicrobial prophylaxis against infective endocarditis in adults and children undergoing interventional procedures. National Institute for Health and Care Excellence. Published March 17, 2008. Updated July 8, 2016.
Accessed September 7, 2023 No relevant cardiac condition Antibiotic prophylaxis isn’t suggested for pts w/o relevant cardiac conditions,1 per AHA. Guidance from other groups differs: - ESC2 suggests considering prophylaxis for cardiac txp pts (w/o specifying valvulopathy). Pts <6mo post-op vascular graft, vena cava filter, or CNS ventriculoatrial shunt are also deemed high risk. For intermediate-risk pts, prophylaxis isn’t routine (no evidence), but consider case-by-case: rheumatic heart dz, nonrheumatic degenerative valve dz, abnl congenital valve (e.g., bicuspid aortic), implanted electronic devices, hypertrophic cardiomyopathy.
- For mod-risk3 pts, JCS weakly recommends considering prophylaxis, case-by-case after discussion w/ pt.
- NICE.4 Antibiotic prophylaxis not routinely recommended for dental procedures; don’t offer chlorhexidine mouthwash as prophylaxis.
Footnotes 1 AHA 2021. Wilson WR, et al. Prevention of Viridans Group Streptococcal Infective Endocarditis: A Scientific Statement From the American Heart Association. Circulation. 2021 May 18;143(20):e963-e978. Full-text article
Prophylaxis not recommended for pacemaker/similar implanted electronics, completed closed septal defect devices, peripheral vascular graft/patch (including for HD), coronary/vascular stents, CNS ventriculoatrial shunt, vena cava filter, pledget.
Four conditions warrant prophylaxis for specific dental/oral surgery procedures:
• Prosthetic cardiac valve/material
◦ Prosthetic valve (including transcatheter-implanted)
◦ Valve repair w/ device (e.g., annuloplasty ring/clip)
◦ LVAD or implantable heart
• Previous/relapsed/recurrent IE
• CHD
◦ Unrepaired cyanotic congenital CHD (including palliative shunt, conduit)
◦ Congenital defect repaired w/ prosthetic material/device (via surgery/transcatheter) w/in 6mo after repair
◦ Repaired w/ residual defects @ site of/adjacent to prosthetic patch/device
◦ Surgical/transcatheter PA valve/conduit
• Cardiac txp w/ valvulopathy
2 ESC 2023. Delgado V, et al. 2023 ESC Guidelines for the management of endocarditis: Developed by the task force on the management of endocarditis of the European Society of Cardiology (ESC). Eur Heart J. Preprint. Posted online August 25, 2023.
Full-text article
3 JCS 2017. Mod risk:
• Non–high-risk CHD including bicuspid aortic valve (however, isolated ASD and repaired VSD/PDA 6+ mo post-op w/o residual shunt deemed low risk)
• MVP w/ regurg
• acquired valvular dz (however, mitral stenosis w/o regurg and functional murmur are deemed low risk)
• hypertrophic cardiomyopathy w/ obstruction
• intracardiac devices (pacemaker, implantable defibrillator)
• chronic central venous catheter
Nakatani S, et al. JCS 2017 Guideline on Prevention and Treatment of Infective Endocarditis. Circ J. 2019 Jul 25;83(8):1767-1809.
Full-text article
4 NICE 2016. Prophylaxis against infective endocarditis: antimicrobial prophylaxis against infective endocarditis in adults and children undergoing interventional procedures. National Institute for Health and Care Excellence. Published March 17, 2008. Updated July 8, 2016.
Accessed September 7, 2023
-
Patient undergoing other procedure type
Antibiotic prophylaxis isn’t recommended for procedures1 other than dental/oral surgery procedures involving gingival tissue/periapical region manipulation or oral mucosal perforation, per AHA.1 Guidance from other groups differs: - ESC2 suggests considering prophylaxis for high-risk2 pts undergoing invasive GI/GU/resp tract, skin, or musculoskeletal procedure, though data limited.
- JCS3 strongly recommends prophylaxis for high-risk3 pts undergoing T&A or pacemaker/cardiac defibrillator implantation. They prefer prophylaxis for invasive procedures on localized infxns, TURP (esp. if prosthetic valve), and CV prosthesis procedures. Only for pts w/ previous IE: prophylaxis recommended for skin incision (esp. if atopic dermatitis), cardiac cath, vaginal delivery/hysterectomy, abortion, IUD removal, urethral dilatation, intrauterine curettage, colonoscopy/proctoscopy, esophageal varices sclerotherapy, esophageal stenosis dilatation, biliary tract surgery.
- NICE.4 Antibiotic prophylaxis not routinely recommended for upper/lower GI tract, GU/obstetric, ENT, or upper/lower resp tract procedures.
Footnotes 1 AHA 2021. Wilson WR, et al. Prevention of Viridans Group Streptococcal Infective Endocarditis: A Scientific Statement From the American Heart Association. Circulation. 2021 May 18;143(20):e963-e978. Full-text article
Dental procedures not warranting prophylaxis include x-rays; anesthetic injection in noninfected tissue; placing removable prosthodontic/orthodontic appliances or orthodontic brackets; orthodontic appliance adjustment; primary teeth shedding or lip/oral mucosa traumatic bleeding.
2 ESC 2023. High-risk = previous IE, surgically implanted prosthetic valve, surgical valve repair w/ any material, transcatheter aortic/pulmonary valve prosthesis, VAD, CHD (untreated cyanotic, palliative shunt/conduit/prosthesis, or <6mo after complete repair), or <6mo after closure device for septal defect/left atrial appendage, vascular graft, vena cava filters, or ventriculoatrial shunt. May consider for pts w/ transcatheter mitral/tricuspid valve repair or cardiac txp.
Delgado V, et al. 2023 ESC Guidelines for the management of endocarditis: Developed by the task force on the management of endocarditis of the European Society of Cardiology (ESC). Eur Heart J. Preprint. Posted online August 25, 2023.
Full-text article
3 JCS 2017. High-risk = previous IE, prosthetic valve/annular ring, CHD (unrepaired cyanotic, residual lesions at repair site w/ patch/materials, <6mo after repair w/ artificial material), or systemic-pulmonary circulation shunt.
Procedures not warranting prophylaxis include:
• Bronchoscopy/laryngoscopy, endotracheal intubation
• Tube insertion for tympanic perforation
• TEE, GI endoscopy
• Urethral catheterization, cystoscopy
• Central venous catheterization
Nakatani S, et al. JCS 2017 Guideline on Prevention and Treatment of Infective Endocarditis. Circ J. 2019 Jul 25;83(8):1767-1809.
Full-text article
4 NICE 2016. Prophylaxis against infective endocarditis: antimicrobial prophylaxis against infective endocarditis in adults and children undergoing interventional procedures. National Institute for Health and Care Excellence. Published March 17, 2008. Updated July 8, 2016.
Accessed September 7, 2023
|