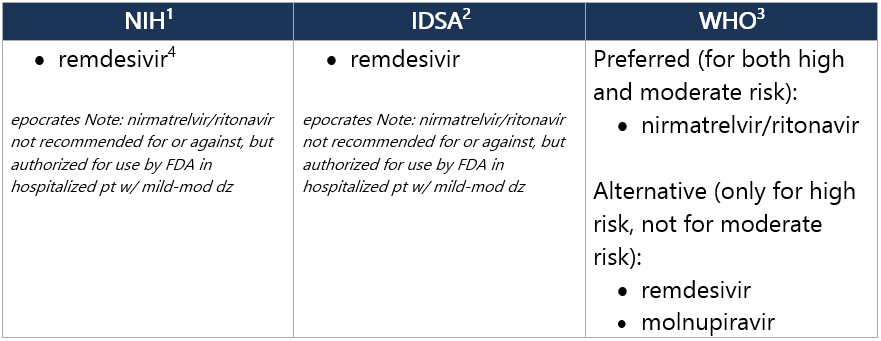
-
Mild-mod dz at LOW risk for progressing to severe dz General mgmt - Minimize risk of SARS-CoV-2 transmission
- Re-evaluate risk for dz progression & need for COVID-19–specific tx
Therapeutic mgmt - Provide supportive care & sx mgmt
- No COVID-19–specific treatments recommended
- Systemic corticosteroids not recommended unless needed for another indication
- Anticoagulation: NOT recommended unless other indication for antithrombotic tx
- Note: Activity of anti–SARS-CoV-2-specific monoclonal antibodies against currently circulating variants is significantly reduced & tx w/ these agents is NOT recommended
Footnotes 1 NIH 2023. COVID-19 Treatment Guidelines Panel. Coronavirus Disease 2019 (COVID-19) Treatment Guidelines. National Institutes of Health. Last update 11/2/23. Accessed 11/13/23
Mild-mod dz at HIGH risk for progressing to severe dz General mgmt - Minimize risk of SARS-CoV-2 transmission
Therapeutic mgmt - Provide supportive care & sx mgmt
- Anticoagulation: NOT recommended unless other indication for antithrombotic tx1
- Note: Activity of anti–SARS-CoV-2-specific monoclonal antibodies against currently circulating variants is significantly reduced & tx w/ these agents is NOT recommended
Footnotes 1 NIH 2023. COVID-19 Treatment Guidelines Panel. Coronavirus Disease 2019 (COVID-19) Treatment Guidelines. National Institutes of Health. Last update 11/2/23. Accessed 11/13/23
2 IDSA 2023. Infectious Diseases Society of America Guidelines on the Treatment and Management of Patients With COVID-19. Last update 6/26/23. Accessed 11/13/23
3 WHO 2023. Therapeutics and COVID-19: Living Guideline. World Health Organization. Last update 1/13/23. Accessed 3/15/23
Pts at high risk of hospitalization include those w/ diagnosed immunodeficiency syndromes, solid organ transplant recipients being treated w/ immunosuppressants, and those w/ autoimmune illness receiving immunosuppressants.
Pts at moderate risk of hospitalization are those >65 yo, those w/ obesity, diabetes, or a chronic condition such as COPD, kidney or liver dz, or cancer, those w/ disabilities, and those w/ comorbidities of chronic dz.
4 NIH 2023. Consider longer or additional courses in immunocompromised pts w/ prolonged symptomatic COVID-19 and evidence of ongoing viral replication despite antiviral tx. -
Mild-mod dz at LOW risk for progressing to severe dz General mgmt - Minimize risk of SARS-CoV-2 transmission
- Re-evaluate risk for dz progression & need for COVID-19–specific tx
Therapeutic mgmt - No COVID-19–specific treatments recommended
- Systemic corticosteroids not recommended unless needed for another indication
- Anticoagulation: Prophylactic-dose heparin recommended unless contraindicated or therapeutic anticoagulation indicated1
- Note: Activity of anti–SARS-CoV-2-specific monoclonal antibodies against currently circulating variants is significantly reduced & tx w/ these agents is NOT recommended
Footnotes 1 NIH 2023. COVID-19 Treatment Guidelines Panel. Coronavirus Disease 2019 (COVID-19) Treatment Guidelines. National Institutes of Health. Last update 11/2/23. Accessed 11/13/23
Mild-mod dz at HIGH risk for progressing to severe dz General mgmt - Minimize risk of SARS-CoV-2 transmission
Therapeutic mgmt - Anticoagulation: Prophylactic-dose heparin recommended unless contraindicated or therapeutic anticoagulation indicated1
- Note: Activity of anti–SARS-CoV-2-specific monoclonal antibodies against currently circulating variants is significantly reduced & tx w/ these agents is NOT recommended
Footnotes 1 NIH 2023. COVID-19 Treatment Guidelines Panel. Coronavirus Disease 2019 (COVID-19) Treatment Guidelines. National Institutes of Health. Last update 11/2/23. Accessed 11/13/23
2 IDSA 2023. Infectious Diseases Society of America Guidelines on the Treatment and Management of Patients With COVID-19. Last update 6/26/23. Accessed 11/13/23
3 WHO 2023. Therapeutics and COVID-19: Living Guideline. World Health Organization. Last update 1/13/23. Accessed 3/15/23
epocrates note: WHO guidelines give recommendations for non-severe dz but don’t differentiate inpts vs outpts; recommendations are extrapolated from outpts.
Pts at high risk of hospitalization include those w/ diagnosed immunodeficiency syndromes, solid organ transplant recipients being treated w/ immunosuppressants, and those w/ autoimmune illness receiving immunosuppressants.
Pts at moderate risk of hospitalization are those >65 yo, those w/ obesity, diabetes, or a chronic condition such as COPD, kidney or liver dz, or cancer, those w/ disabilities, and those w/ comorbidities of chronic dz.
4 NIH 2023. Consider longer or additional courses in immunocompromised pts w/ prolonged symptomatic COVID-19 and evidence of ongoing viral replication despite antiviral tx. Severe dz but not critically ill General mgmt - Minimize risk of SARS-CoV-2 transmission
Therapeutic mgmt - Anticoagulation: If D-dimer >ULN & no incr bleeding risk, therapeutic-dose heparin recommended for nonpregnant pts; otherwise, prophylactic-dose heparin recommended unless contraindicated or therapeutic anticoagulation indicated1
- Note: Activity of anti–SARS-CoV-2-specific monoclonal antibodies against currently circulating variants is significantly reduced & tx w/ these agents is NOT recommended
Footnotes 1 NIH 2023. COVID-19 Treatment Guidelines Panel. Coronavirus Disease 2019 (COVID-19) Treatment Guidelines. National Institutes of Health. Last update 11/2/23. Accessed 11/13/23
2 IDSA 2023. Infectious Diseases Society of America Guidelines on the Treatment and Management of Patients With COVID-19. Last update 6/26/23. Accessed 11/13/23
3 WHO 2023. Therapeutics and COVID-19: Living Guideline. World Health Organization. Last update 1/13/23. Accessed 3/15/23
4 Equivalent daily glucocorticoid dose may be substituted if dexamethasone unavailable.
5 If dexamethasone contraindicated, consider remdesivir + baricitinib.
6 If none of the preferred or alternative options are available or feasible to use, may use tofacitinib or sarilumab IV.
7 If neither baricitinib nor tocilizumab is available or feasible to use, may use tofacitinib as an alternative to baricitinib, & sarilumab IV as an alternative to tocilizumab IV.
8 If neither baricitinib nor IL-6 receptor blocker is available, may use ruxolitinib or tofacitinib. Studies used ruxolitinib 5 mg PO bid x14 days. Critically ill, needs non-invasive ventilation or high-flow nasal cannula General mgmt - Minimize risk of SARS-CoV-2 transmission
Therapeutic mgmt - Anticoagulation: Prophylactic-dose heparin recommended unless contraindicated or therapeutic anticoagulation indicated; if started on therapeutic-dose heparin prior to ICU, switch to prophylactic-dose heparin unless therapeutic anticoagulation indicated1
- Note: Activity of anti–SARS-CoV-2-specific monoclonal antibodies against currently circulating variants is significantly reduced & tx w/ these agents is NOT recommended
Footnotes 1 NIH 2023. COVID-19 Treatment Guidelines Panel. Coronavirus Disease 2019 (COVID-19) Treatment Guidelines. National Institutes of Health. Last update 11/2/23. Accessed 11/13/23
2 IDSA 2023. Infectious Diseases Society of America Guidelines on the Treatment and Management of Patients With COVID-19. Last update 6/26/23. Accessed 11/13/23
3 WHO 2023. Therapeutics and COVID-19: Living Guideline. World Health Organization. Last update 1/13/23. Accessed 3/15/23
4 Equivalent daily glucocorticoid dose may be substituted if dexamethasone unavailable.
5 If none of the preferred or alternative options are available or feasible to use, may use tofacitinib or sarilumab IV.
6 If neither baricitinib nor tocilizumab is available or feasible to use, may use tofacitinib as an alternative to baricitinib, & sarilumab IV as an alternative to tocilizumab IV.
7 If neither baricitinib nor IL-6 receptor blocker is available, may use ruxolitinib or tofacitinib. Studies used ruxolitinib 5 mg PO bid x14 days. Critically ill, needs invasive mech vent or ECMO General mgmt - Minimize risk of SARS-CoV-2 transmission
Therapeutic mgmt - Anticoagulation: Prophylactic-dose heparin recommended unless contraindicated or therapeutic anticoagulation indicated; if started on therapeutic-dose heparin prior to ICU, switch to prophylactic-dose heparin unless therapeutic anticoagulation indicated1
- Note: Activity of anti–SARS-CoV-2-specific monoclonal antibodies against currently circulating variants is significantly reduced & tx w/ these agents is NOT recommended
Footnotes 1 NIH 2023. COVID-19 Treatment Guidelines Panel. Coronavirus Disease 2019 (COVID-19) Treatment Guidelines. National Institutes of Health. Last update 11/2/23. Accessed 11/13/23
2 IDSA 2023. Infectious Diseases Society of America Guidelines on the Treatment and Management of Patients With COVID-19. Last update 6/26/23. Accessed 11/13/23
3 WHO 2023. Therapeutics and COVID-19: Living Guideline. World Health Organization. Last update 1/13/23. Accessed 3/15/23
4 Equivalent daily glucocorticoid dose may be substituted if dexamethasone unavailable.
5 If neither baricitinib nor tocilizumab is available or feasible to use, may use tofacitinib as an alternative to baricitinib, & sarilumab IV as an alternative to tocilizumab IV.
6 If neither baricitinib nor IL-6 receptor blocker is available, may use ruxolitinib or tofacitinib. Studies used ruxolitinib 5 mg PO bid x14 days.
|