-
Dyspnea, chronic cough/sputum, and/or COPD risk factors
Confirm dx w/ spirometry pre/post bronchodilator in pts >40 yo w/ chronic sx and/or exposure risk factors. If possible, test for SARS-CoV-2 prior to spirometry/PFTs and delay if test is positive. If COPD confirmed, perform complete assessment (see below). - Hx: progressive/persistent dyspnea, worse w/ exercise. Chronic cough (may be intermittent, unproductive, or absent) w/ or w/o chronic sputum (any pattern). Sx may vary day to day. Dyspnea/cough/sputum may precede airflow limitation by yrs. Airflow limitation may exist w/o dyspnea/cough/sputum and vice versa. May have recurrent wheeze/chest tightness, recurrent lower resp infxns, fatigue, wt loss/anorexia.
- Risks: smoke (tobacco, heating/cooking fuel), occupational (dust, chemicals, fumes), genetic, developmental abnormalities, low birth wt, prematurity, childhood resp infxn.
- Exam: Exam rarely (if ever) diagnostic. Wheeze doesn’t confirm asthma; absence doesn’t exclude COPD. Ankle swelling may signal cor pulmonale.
- Post-BD FEV1/FVC <0.7 required to confirm COPD; if initial ratio 0.6 to 0.8 post BD, recheck on separate occasion. Avoid PEF due to low specificity. Degree of reversibility pre/post BD not shown to improve COPD dx, differentiate from asthma, or predict tx response. Significant lung pathology can be present w/o airflow limitation.
◦ Lung volumes help characterize severity but aren’t essential to pt mgmt.
◦ If sx (dyspnea) out of proportion to degree of airflow obstruction, measure DLco.
DDx of chronic cough: asthma, lung CA, TB, bronchiectasis, left HF, ILD, CF, idiopathic, chronic allergic rhinitis, postnasal drip syndrome, upper airway cough syndrome, GERD, ACE inhibitors. DDx of COPD: asthma, CHF, bronchiectasis, TB, obliterative bronchiolitis, diffuse panbronchiolitis. Sx, risk factors, CXR/HRCT may help distinguish. Once dx confirmed by spirometry, initial assessment includes: - Severity of airflow obstruction (GOLD 1/mild: FEV1 ≥80% predicted; GOLD 2/mod: FEV1 50% to <80% predicted; GOLD 3/severe: FEV1 30% to <50% predicted; GOLD 4/very severe: FEV1 <30% predicted).
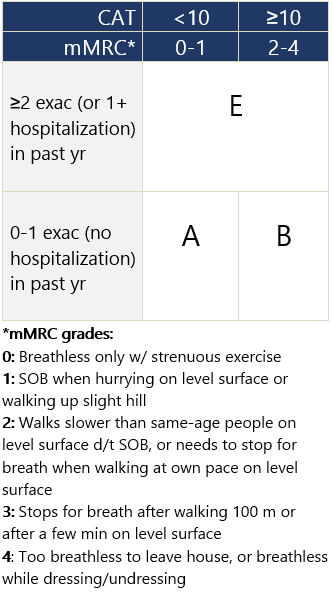
- Nature and current magnitude of sx w/ validated scale: CAT (preferred) or mMRC* scale + exac risk.
- Hx of mod/severe exac: number of exac and hospitalizations.
- Blood eosinophil count: guides ICS use, may predict exac risk.
- Comorbidities: CV dz, skeletal muscle dysfxn, metabolic syndrome, osteoporosis, depression, anxiety, lung CA. Pseudomonas aeruginosa colonization independently ↑ risk of hospitalization and all-cause mortality.
- Screen for AATD once or if +FHx, esp. in areas w/ high AATD prevalence (Scandinavia, White northern Europeans, absent from Asian/Black populations).1
If discordance between sx and level of airflow obstruction, consider additional tests and impact of comorbidities (e.g., IHD). - Lung volumes: Test by body plethysmography. Can characterize severity but not essential to mgmt.
- DLco: Measure in anyone w/ dyspnea disproportionate to airflow obstruction by spirometry. DLco <60% predicted assoc w/ ↑sx, ↓exercise capacity, worse health status, ↑risk of death.
- SpO2 and ABG: Check in all w/ signs suggesting resp failure. Guides need for supplemental O2. If SpO2 <92%, check ABG.
- Exercise testing: may reveal degree of impairment and need for rehab, assesses disability. Predicts prognosis. Use shuttle walk test or self-paced 6-min walk test. Physical activity monitoring may be more relevant to prognosis than exercise capacity eval.
- Imaging: CXR not useful for dx but may exclude alternative dx or establish resp/CV/skeletal comorbidities. Consider chest CT for pts w/ persistent exac, sx out of proportion to dz severity on lung fxn testing, FEV1 <45% predicted w/ significant hyperinflation/gas trapping, or if lung CA screening criteria are met.
- BODE (Body mass index, Obstruction, Dyspnea, and Exercise) score may be predictive of survival.
GOLD COPD group based on mod/severe exacerbations and sx scores Footnotes 1 α 1-Antitrypsin deficiency: Memorandum from a WHO meeting. Bulletin of the World Health Organization. 1997;75:397-415. PDF -
No dyspnea, no chronic cough/sputum, no COPD risk factors
General population screening is controversial. USPSTF recommends against screening asymptomatic adults w/o risk factors. GOLD advocates active case-finding, not screening spirometry. - Asymptomatic pts w/o significant exposures probably don’t warrant spirometry. If pt has risks (e.g., >20 pack/yr smoking, recurrent chest infxns), consider spirometry for early case-finding.
- If pt undergoing LDCT for lung CA screening, or has incidental lung imaging abnormalities (emphysema, air trapping, airway wall thickening, mucus plugging), GOLD recommends spirometry to screen for COPD.
- Identifying pts before significant sx develop not shown to improve COPD outcomes or impact mgmt.
- If family member w/ A1AT deficiency: A1AT screening/counseling; classic pt: <45 yo w/ panlobular basal emphysema.
|